"Lupus" redirects here. For other uses, see Lupus (disambiguation).
| Systemic lupus erythematosus | |
|---|---|
| Classification and external resources | |
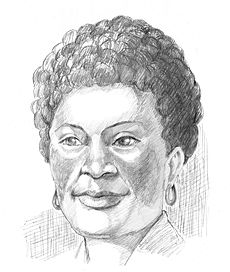
Drawing of the typical "butterfly rash" found in lupus
| |
| ICD-10 | M32 |
| ICD-9 | 710.0 |
| OMIM | 152700 |
| DiseasesDB | 12782 |
| MedlinePlus | 000435 |
| eMedicine | med/2228 emerg/564 |
| Patient UK | Systemic lupus erythematosus |
| MeSH | D008180 |
Systemic lupus erythematosus  i/sɪˈstɛmɪk ˈluːpəs ˌɛrɪθiːməˈtoʊsəs/, often abbreviated as SLE or lupus, is asystemic autoimmune disease (or autoimmune connective tissue disease) in which the body’s immune system mistakenly attacks normal, healthy tissue. This can affect the skin, joints, kidneys, brain, and other organs. And can result in symptoms, such as inflammation and swelling. When the immune system is functioning normally, it makes proteins called antibodies that protect and fight against antigens, like viruses and bacteria. Lupus causes the immune system to be unable to differentiate antigens and healthy tissue. As a result, the immune system directs antibodies against healthy tissue, which causes swelling, pain, and tissue damage. Although the underlying cause of autoimmune diseases is unknown, most doctors believe that lupus results from both genetic and environmental stimuli.
i/sɪˈstɛmɪk ˈluːpəs ˌɛrɪθiːməˈtoʊsəs/, often abbreviated as SLE or lupus, is asystemic autoimmune disease (or autoimmune connective tissue disease) in which the body’s immune system mistakenly attacks normal, healthy tissue. This can affect the skin, joints, kidneys, brain, and other organs. And can result in symptoms, such as inflammation and swelling. When the immune system is functioning normally, it makes proteins called antibodies that protect and fight against antigens, like viruses and bacteria. Lupus causes the immune system to be unable to differentiate antigens and healthy tissue. As a result, the immune system directs antibodies against healthy tissue, which causes swelling, pain, and tissue damage. Although the underlying cause of autoimmune diseases is unknown, most doctors believe that lupus results from both genetic and environmental stimuli.
There are many kinds of lupus. The most common type is systemic lupus erythematosus (SLE), which affects many internal organs in the body. SLE most often harms the heart, joints, skin, lungs, blood vessels, liver, kidneys, andnervous system. The course of the disease is unpredictable, with periods of illness (called flares) alternating withremissions. The disease occurs nine times more often in women than in men, especially in women in child-bearing years ages 15 to 35, and is also more common in those of non-European descent.[1][2][3]
While there is no cure for SLE. It is treated with immunosuppression, mainly with cyclophosphamide, corticosteroids and other immunosuppressants. The goal of these treatments is to keep symptoms under control. SLE can be fatal. The leading cause of death is from cardiovascular disease due to accelerated atherosclerosis. Survival for people with SLE in the United States, Canada, and Europe has risen to approximately 95% at five years, 90% at 10 years, and 78% at 20 years,[1] and now approaches that of matched controls without lupus.
Childhood systemic lupus erythematosus generally presents between the ages of 3 and 15, with girls outnumbering boys 4:1, and typical skin manifestations being butterfly eruption on the face and photosensitivity.[4]
Lupus is Latin for wolf. In the 18th century, when lupus was just starting to be recognized as a disease, it was thought that it was caused by the bite of a wolf.[5] This may have been because of the distinctive rash characteristic of lupus. (Once full-blown, the round, disk-shaped rashes heal from the inside out, leaving a bite-like imprint.)
Signs and symptoms
SLE is one of several diseases known as "the great imitators" because it often mimics or is mistaken for other illnesses.[7]SLE is a classical item in differential diagnosis,[2] because SLE symptoms vary widely and come and go unpredictably. Diagnosis can thus be elusive, with some people suffering unexplained symptoms of untreated SLE for years.
Common initial and chronic complaints include fever, malaise, joint pains, myalgias, fatigue, and temporary loss of cognitive abilities. Because they are so often seen with other diseases, these signs and symptoms are not part of the diagnostic criteria for SLE. When occurring in conjunction with other signs and symptoms (see below), however, they are considered suggestive.[8]
While SLE can occur in both males and females, the symptoms associated with each sex are different. Females tend to have greater relapses, leukopenia, more arthritis, Raynaud phenomenon, and psychiatric symptoms. On the other hand, males tend to have more skin problems, seizures, renal disease serositis, and peripheral neuropathy.[9]
Skin
As many as 70% of people with lupus have some dermatological symptoms. The three main categories of lesions are chronic cutaneous [discoid] lupus, subacute cutaneous lupus, and acute cutaneous lupus. Sufferers of discoid lupus may exhibit thick, red scaly patches on the skin. Similarly, subacute cutaneous lupus manifests as red, scaly patches of skin but with distinct edges. Acute cutaneous lupus manifests as a rash. Some suffer from the classic malar rash (or butterfly rash) associated with the disease.[10] Hair loss, mouth, nasal, urinary tract, and vaginal ulcers, and lesions on the skin are other possible manifestations. Tiny tears in the delicate tissue around the eyes can occur after even minimal rubbing.
Muscles and bones
The most commonly sought medical attention is for joint pain, with the small joints of the hand and wrist usually affected, although all joints are at risk. The Lupus Foundation of America estimates more than 90 percent of those affected will experience joint and/or muscle pain at some time during the course of their illness.[11] Unlike rheumatoid arthritis, lupus arthritis is less disabling and usually does not cause severe destruction of the joints. Fewer than ten percent of people with lupus arthritis will develop deformities of the hands and feet.[11] SLE patients are at particular risk of developing osteoarticulartuberculosis.[12]
A possible association between rheumatoid arthritis and SLE has been suggested,[13] and SLE may be associated with an increased risk of bone fractures in relatively young women.[14]
Blood
Anemia is common in children with SLE[15] and develops in about 50% of cases.[16] Low platelet and white blood cell counts may be due to the disease or a side effect of pharmacological treatment. People with SLE may have an association with antiphospholipid antibody syndrome[17] (a thrombotic disorder), wherein autoantibodies to phospholipids are present in their serum. Abnormalities associated with antiphospholipid antibody syndrome include a paradoxical prolongedpartial thromboplastin time (which usually occurs in hemorrhagic disorders) and a positive test for antiphospholipid antibodies; the combination of such findings have earned the term "lupus anticoagulant-positive". Another autoantibody finding in SLE is the anticardiolipin antibody, which can cause a false positive test forsyphilis.[citation needed]
Heart
A person with SLE may have inflammation of various parts of the heart, such as pericarditis, myocarditis, and endocarditis. The endocarditis of SLE is characteristically noninfective (Libman-Sacks endocarditis), and involves either the mitral valve or the tricuspid valve. Atherosclerosis also tends to occur more often and advances more rapidly than in the general population.[18][19][20]
Lungs
Lung and pleura inflammation can cause pleuritis, pleural effusion, lupus pneumonitis, chronic diffuse interstitial lung disease, pulmonary hypertension, pulmonary emboli, pulmonary hemorrhage, and shrinking lung syndrome.
Kidneys
Painless hematuria or proteinuria may often be the only presenting renal symptom. Acute or chronic renal impairment may develop with lupus nephritis, leading to acute or end-stage renal failure. Because of early recognition and management of SLE, end-stage renal failure occurs in less than 5%[citation needed] of cases.
A histological hallmark of SLE is membranous glomerulonephritis with "wire loop" abnormalities.[21] This finding is due to immune complex deposition along theglomerular basement membrane, leading to a typical granular appearance in immunofluorescence testing.
Neuropsychiatric
Neuropsychiatric syndromes can result when SLE affects the central or peripheral nervous systems. The American College of Rheumatology defines 19 neuropsychiatric syndromes in systemic lupus erythematosus.[22] The diagnosis of neuropsychiatric syndromes concurrent with SLE is one of the most difficult challenges in medicine, because it can involve so many different patterns of symptoms, some of which may be mistaken for signs of infectious disease or stroke.[23]
The most common neuropsychiatric disorder people with SLE have is headache,[24] although the existence of a specific lupus headache and the optimal approach to headache in SLE cases remains controversial.[25] Other common neuropsychiatric manifestation of SLE include cognitive dysfunction, mood disorder,cerebrovascular disease,[24] seizures, polyneuropathy,[24] anxiety disorder, and psychosis. It can rarely present with intracranial hypertension syndrome, characterized by an elevated intracranial pressure, papilledema, and headache with occasional abducens nerve paresis, absence of a space-occupying lesion or ventricular enlargement, and normal cerebrospinal fluid chemical and hematological constituents.[26]
More rare manifestations are acute confusional state, Guillain-Barré syndrome, aseptic meningitis, autonomic disorder, demyelinating syndrome, mononeuropathy(which might manifest as mononeuritis multiplex), movement disorder (more specifically, chorea), myasthenia gravis, myelopathy, cranial neuropathy and plexopathy.
Neurological
Neural symptoms contribute to a significant percentage of morbidity and mortality in patients with lupus.[27] As a result, the neural side of lupus is being studied in hopes of reducing morbidity and mortality rates.[22] The neural manifestation of lupus is known as neuropsychiatric systemic lupus erythematosus (NPSLE). One aspect of this disease is severe damage to the epithelial cells of the blood–brain barrier.
Lupus has a wide range of symptoms which span the body. The neurological symptoms include headaches,[24] depression, seizures, cognitive dysfunction, mood disorder, cerebrovascular disease,[24] polyneuropathy,[24] anxiety disorder, psychosis, and in some extreme cases, personality disorders.[28] In certain regions, depression reportedly affects up to 60% of women suffering from SLE.[29]
Reproductive
Further information: Systemic lupus erythematosus and pregnancy
SLE causes an increased rate of fetal death in utero and spontaneous abortion (miscarriage). The overall live-birth rate in SLE patients has been estimated to be 72%.[30] Pregnancy outcome appears to be worse in SLE patients whose disease flares up during pregnancy.[31]
Neonatal lupus is the occurrence of SLE symptoms in an infant born from a mother with SLE, most commonly presenting with a rash resembling discoid lupus erythematosus, and sometimes with systemic abnormalities such as heart block or hepatosplenomegaly.[32] Neonatal lupus is usually benign and self-limited.[32]
Systemic
Fatigue in SLE is probably multifactorial and has been related to not only disease activity or complications such as anemia or hypothyroidism, but also to pain,depression, poor sleep quality, poor physical fitness and lack of social support.[33][34]
Causes
There is no one specific cause of SLE. There are, however, a number of environmental triggers and a number of genetic susceptibilities.[35][36]
Genetics
The first mechanism may arise genetically. Research indicates SLE may have a genetic link. SLE does run in families, but no single causal gene has been identified. Instead, multiple genes appear to influence a person's chance of developing lupus when triggered by environmental factors. HLA class I, class II, and class III genes are associated with SLE, but only classes I and II contribute independently to increased risk of SLE.[37] Other genes which contain risk variants for SLE are IRF5,PTPN22, STAT4,[38] CDKN1A,[39] ITGAM, BLK,[38] TNFSF4 and BANK1.[40] Some of the susceptibility genes may be population specific.[38]
Drug reactions
Drug-induced lupus erythematosus is a (generally) reversible condition that usually occurs in people being treated for a long-term illness. Drug-induced lupus mimics SLE. However, symptoms of drug-induced lupus generally disappear once the medication that triggered the episode is stopped. More than 38 medications can cause this condition, the most common of which are procainamide, isoniazid, hydralazine, quinidine, and phenytoin.[2]
Non-systemic forms of lupus
Discoid (cutaneous) lupus is limited to skin symptoms and is diagnosed by biopsy of rash on the face, neck, scalp or arms. Approximately 5% of patients with DLE progress to SLE.[41]
Pathophysiology
One manifestation of SLE is abnormalities in apoptosis, a type of programmed cell death in which aging or damaged cells are neatly disposed of as a part of normal growth or functioning.
In SLE, the body's immune system produces antibodies against itself, particularly against proteins in the cell nucleus. SLE is triggered by environmental factors that are unknown.
The immune system must balance between being sensitive enough to protect against infection, and becoming sensitized to attack the body's own proteins (autoimmunity). During an immune reaction to a foreign stimulus, such as bacteria, virus, or allergen, immune cells that would normally be deactivated due to their affinity for self tissues can be abnormally activated by signaling sequences of antigen-presenting cells. Thus triggers may include viruses, bacteria, allergens (IgEand other hypersensitivity), and can be aggravated by environmental stimulants such as ultraviolet light and certain drug reactions. These stimuli begin a reaction that leads to destruction of other cells in the body and exposure of their DNA, histones, and other proteins, particularly parts of the cell nucleus. The body's sensitized B-lymphocyte cells will now produce antibodies against these nuclear-related proteins. These antibodies clump into antibody-protein complexes which stick to surfaces and damage blood vessels in critical areas of the body, such as the glomeruli of the kidney; these antibody attacks are the cause of SLE. Researchers are now identifying the individual genes, the proteins they produce, and their role in the immune system. Each protein is a link on the autoimmune chain, and researchers are trying to find drugs to break each of those links.[2][42][43]
SLE is a chronic inflammatory disease believed to be a type III hypersensitivity response with potential type II involvement.[44] Reticulate and stellate acral pigmentation should be considered a possible manifestation of SLE and high titers of anti-cardiolipin antibodies, or a consequence of therapy.[45]
Abnormalities in cell death signaling
- Apoptosis is increased in monocytes and keratinocytes
- Expression of Fas by B cells and T cells is increased
- There are correlations between the apoptotic rates of lymphocytes and disease activity.
- Necrosis is increased in T lymphocytes.
Tingible body macrophages (TBMs) – large phagocytic cells in the germinal centers of secondary lymph nodes – express CD68 protein. These cells normally engulf B cells that have undergone apoptosis after somatic hypermutation. In some people with SLE, significantly fewer TBMs can be found, and these cells rarely contain material from apoptotic B cells. Also, uningested apoptotic nuclei can be found outside of TBMs. This material may present a threat to the tolerization of B cells and T cells. Dendritic cells in the germinal center may endocytose such antigenic material and present it to T cells, activating them. Also, apoptotic chromatin and nuclei may attach to the surfaces of follicular dendritic cells and make this material available for activating other B cells that may have randomly acquired self-specificity through somatic hypermutation.[46] Necrosis, a pro-inflammatory form of cell death, is increased in T lymphocytes, due to mitochondrial dysfunction, oxidative stress, and depletion of ATP.[47]
Clearance deficiency
Impaired clearance of dying cells is a potential pathway for the development of this systemic autoimmune disease. This includes deficient phagocytic activity and scant serum components in addition to increased apoptosis.
Monocytes isolated from whole blood of SLE sufferers show reduced expression of CD44 surface molecules involved in the uptake of apoptotic cells. Most of the monocytes and tingible body macrophages (TBMs), which are found in the germinal centres of lymph nodes, even show a definitely different morphology; they are smaller or scarce and die earlier. Serum components like complement factors, CRP, and some glycoproteins are, furthermore, decisively important for an efficiently operating phagocytosis. With SLE, these components are often missing, diminished, or inefficient.
Recent research has found an association between certain lupus patients (especially those with lupus nephritis) and an impairment in degrading neutrophil extracellular traps (NETs). These were due to DNAse1 inhibiting factors, or NET protecting factors in patient serum, rather than abnormalities in the DNAse1 itself.[48] DNAse1 mutations in lupus have so far only been found in some Japanese cohorts.[49]
The clearance of early apoptotic cells is an important function in multicellular organisms. It leads to a progression of the apoptosis process and finally to secondary necrosis of the cells if this ability is disturbed. Necrotic cells release nuclear fragments as potential autoantigens, as well as internal danger signals, inducing maturation of dendritic cells (DCs), since they have lost their membranes' integrity. Increased appearance of apoptotic cells also simulates inefficient clearance. That leads to maturation of DCs and also to the presentation of intracellular antigens of late apoptotic or secondary necrotic cells, via MHC molecules. Autoimmunity possibly results by the extended exposure to nuclear and intracellular autoantigens derived from late apoptotic and secondary necrotic cells. B and T cell tolerance for apoptotic cells is abrogated, and the lymphocytes get activated by these autoantigens; inflammation and the production of autoantibodies by plasma cells is initiated. A clearance deficiency in the skin for apoptotic cells has also been observed in people with cutaneous lupus erythematosus (CLE).[50]
Accumulation in germinal centers
In healthy conditions, apoptotic lymphocytes are removed in germinal centres (GC) by specialized phagocytes, the tingible body macrophages (TBM), which is why no free apoptotic and potential autoantigenic material can be seen. In some people with SLE, accumulation of apoptotic debris can be observed in GC because of an ineffective clearance of apoptotic cells. In close proximity to TBM, follicular dendritic cells (FDC) are localised in GC, which attach antigen material to their surface and, in contrast to bone marrow-derived DC, neither take it up nor present it via MHC molecules.
Autoreactive B cells can accidentally emerge during somatic hypermutation and migrate into the germinal center light zone. Autoreactive B cells, maturated coincidentally, normally do not receive survival signals by antigen planted on follicular dendritic cells, and perish by apoptosis. In the case of clearance deficiency, apoptotic nuclear debris accumulates in the light zone of GC and gets attached to FDC. This serves as a germinal centre survival signal for autoreactive B-cells. After migration into the mantle zone, autoreactive B cells require further survival signals from autoreactive helper T cells, which promote the maturation of autoantibody-producing plasma cells and B memory cells. In the presence of autoreactive T cells, a chronic autoimmune disease may be the consequence.
Anti-nRNP autoimmunity
Autoantibodies to nRNP A and nRNP C initially targeted restricted, proline-rich motifs. Antibody binding subsequently spread to other epitopes. The similarity and cross-reactivity between the initial targets of nRNP and Sm autoantibodies identifies a likely commonality in cause and a focal point for intermolecular epitope spreading.[51]
Others
Elevated expression of HMGB1 was found in the sera of patients and mice with systemic lupus erythematosus, high mobility group box 1 (HMGB1) is a nuclear proteinparticipating in chromatin architecture and transcriptional regulation. Recently, there is increasing evidence HMGB1 contributes to the pathogenesis of chronicinflammatory and autoimmune diseases due to its proinflammatory and immunostimulatory properties.[52]
Diagnosis
Laboratory tests
Antinuclear antibody (ANA) testing and anti-extractable nuclear antigen (anti-ENA) form the mainstay of serologic testing for SLE. Several techniques are used to detect ANAs. Clinically the most widely used method is indirect immunofluorescence (IF). The pattern of fluorescence suggests the type of antibody present in the patient's serum. Direct immunofluorescence can detect deposits of immunoglobulins and complement proteins in the patient's skin. When skin not exposed to the sun is tested, a positive direct IF (the so-called lupus band test) is an evidence of systemic lupus erythematosus.[53]
ANA screening yields positive results in many connective tissue disorders and other autoimmune diseases, and may occur in normal individuals. Subtypes of antinuclear antibodies include anti-Smith and anti-double stranded DNA (dsDNA) antibodies (which are linked to SLE) and anti-histone antibodies (which are linked to drug-induced lupus). Anti-dsDNA antibodies are highly specific for SLE; they are present in 70% of cases, whereas they appear in only 0.5% of people without SLE.[2] The anti-dsDNA antibody titers also tend to reflect disease activity, although not in all cases.[2] Other ANA that may occur in people with SLE areanti-U1 RNP (which also appears in systemic sclerosis and mixed connective tissue disease), SS-A (or anti-Ro) and SS-B (oranti-La; both of which are more common in Sjögren's syndrome). SS-A and SS-B confer a specific risk for heart conduction block in neonatal lupus.[54]
Other tests routinely performed in suspected SLE are complement system levels (low levels suggest consumption by the immune system), electrolytes and renal function (disturbed if the kidney is involved), liver enzymes, and complete blood count.
The lupus erythematosus (LE) cell test was commonly used for diagnosis, but it is no longer used because the LE cells are only found in 50–75% of SLE cases, and they are also found in some people with rheumatoid arthritis, scleroderma, and drug sensitivities. Because of this, the LE cell test is now performed only rarely and is mostly of historical significance.[55]
Diagnostic criteria
Some physicians make a diagnosis on the basis of the American College of Rheumatology (ACR) classification criteria. The criteria, however, were established mainly for use in scientific research including use in randomized controlled trials which require higher confidence levels, so many people with SLE may not pass the full criteria.
Criteria
The American College of Rheumatology (ACR) established eleven criteria in 1982,[56] which were revised in 1997[57] as a classificatory instrument to operationalise the definition of SLE in clinical trials. They were not intended to be used to diagnose individuals and do not do well in that capacity. For the purpose of identifying patients for clinical studies, a person has SLE if any 4 out of 11 symptoms are present simultaneously or serially on two separate occasions. Useful mnemonic for remembering the diagnostic findings or symptoms of SLE is SOAP BRAIN MD (S=serositis, O=oral ulcers, A=arthritis, P=photosensitivity, pulmonary fibrosis, B=blood cells, R=renal, Raynauds, A=ANA, I=immunologic (anti-Sm, anti-dsDNA), N=neuropsych, M=malar rash, D=discoid rash), however, not in order of diagnostic importance.
- Malar rash (rash on cheeks); sensitivity = 57%; specificity = 96%.[58]
- Discoid rash (red, scaly patches on skin that cause scarring); sensitivity = 18%; specificity = 99%.[58]
- Serositis: Pleurisy (inflammation of the membrane around the lungs) or pericarditis (inflammation of the membrane around the heart); sensitivity = 56%;specificity = 86% (pleural is more sensitive; cardiac is more specific).[58]
- Oral ulcers (includes oral or nasopharyngeal ulcers); sensitivity = 27%; specificity = 96%.[58]
- Arthritis: nonerosive arthritis of two or more peripheral joints, with tenderness, swelling, or effusion; sensitivity = 86%; specificity = 37%.[58]
- Photosensitivity (exposure to ultraviolet light causes rash, or other symptoms of SLE flareups); sensitivity = 43%; specificity = 96%.[58]
- Blood—hematologic disorder—hemolytic anemia (low red blood cell count) or leukopenia (white blood cell count<4000 a="" class="mw-redirect" href="http://en.wikipedia.org/wiki/Lymphopenia" l="" nbsp="" style="background: none; color: #0b0080; text-decoration: none;" title="Lymphopenia">lymphopenia
Other than the ACR criteria, people with lupus may also have:[61]
- fever (over 100 °F/ 37.7 °C)
- extreme fatigue
- hair loss
- fingers turning white and/or blue when cold (Raynaud's phenomenon)
Criteria for individual diagnosis
Some people, especially those with antiphospholipid syndrome, may have SLE without four of the above criteria, and also SLE may present with features other than those listed in the criteria.[62][63][64]
Recursive partitioning has been used to identify more parsimonious criteria.[58] This analysis presented two diagnostic classification trees:
- Simplest classification tree: SLE is diagnosed if a person has an immunologic disorder (anti-DNA antibody, anti-Smith antibody, false positive syphilis test, or LE cells) or malar rash. It has sensitivity = 92% and specificity = 92%.
- Full classification tree: Uses 6 criteria. It has sensitivity = 97% and specificity = 95%.
Other alternative criteria have been suggested, e.g. the St. Thomas' Hospital "alternative" criteria in 1998.[65]
Prevention
SLE cannot be prevented, but the consequences can be prevented.[66]
As longevity of people with SLE increases, the likelihood of complications also increases in four areas: cardiovascular disease, infections, osteoporosis, and cancer. Standard preventive measures, screening for related diseases may be necessary to deal with the increased risks due to the side effects of medications. Extra vigilance is considered warranted in particular for cancers affecting the immune system.[67]
Treatment
The treatment of SLE involves preventing flares and reducing their severity and duration when they occur.
Treatment can include corticosteroids and anti-malarial drugs. Certain types of lupus nephritis such as diffuse proliferative glomerulonephritis require bouts of cytotoxic drugs. These drugs include cyclophosphamide and mycophenolate.
Hydroxychloroquine (HCQ) was approved by the FDA for lupus in 1955.[68] Some drugs approved for other diseases are used for SLE 'off-label'. In November 2010, an FDA advisory panel recommended approving belimumab (Benlysta) as a treatment for the pain and flare-ups common in lupus. The drug was approved by the FDA in March 2011.[69][70] N-acetylcysteine has been shown to reverse depletion of the natural anti-oxidant glutathione and to safely improve disease activity in a double-blind placebo-controlled pilot study.[71]
Medications
Due to the variety of symptoms and organ system involvement with SLE, its severity in an individual must be assessed in order to successfully treat SLE. Mild or remittent disease may, sometimes, be safely left untreated. If required, nonsteroidal anti-inflammatory drugs and antimalarials may be used. Medications such as Prednisone, Cellcept and Prograf have been used in the past. A number of potential treatments are in clinical trials.[72]
Disease-modifying antirheumatic drugs
Disease-modifying antirheumatic drugs (DMARDs) are used preventively to reduce the incidence of flares, the process of the disease, and lower the need for steroid use; when flares occur, they are treated with corticosteroids. DMARDs commonly in use are antimalarials such as Plaquenil and immunosuppressants (e.g.methotrexate and azathioprine). Plaquenil (hydroxychloroquine) is an FDA-approved antimalarial used for constitutional, cutaneous, and articular manifestations. Hydroxychloroquine has relatively few side effects, and there is evidence that it improves survival among people who have SLE.[68] Cyclophosphamide is used for severe glomerulonephritis or other organ-damaging complications. Mycophenolic acid is also used for treatment of lupus nephritis, but it is not FDA-approved for this indication, and FDA is investigating reports that it may be associated with birth defects when used by pregnant women.[73]
Immunosuppressive drugs
In more severe cases, medications that modulate the immune system (primarily corticosteroids and immunosuppressants) are used to control the disease and prevent recurrence of symptoms (known as flares). Depending on the dosage, people who require steroids may develop Cushing's syndrome, symptoms of which may include obesity, puffy round face, diabetes mellitus, increased appetite, difficulty sleeping and osteoporosis. These may subside if and when the large initial dosage is reduced, but long-term use of even low doses can cause elevated blood pressure and cataracts.
Numerous new immunosuppressive drugs are being actively tested for SLE. Rather than suppressing the immune system nonspecifically, as corticosteroids do, they target the responses of individual [types of] immune cells. Some of these drugs are already FDA-approved for treatment of rheumatoid arthritis.[68] See alsoBelimumab and Atacicept. Lupuzor has given encouraging results in a phase IIb trial.[74]
Analgesia
Since a large percentage of people with SLE suffer from varying amounts of chronic pain, stronger prescription analgesics (pain killers) may be used if over-the-counter drugs (mainly nonsteroidal anti-inflammatory drugs) do not provide effective relief. Potent NSAIDs such as indomethacin and diclofenac are relatively contraindicated for patients with SLE because they increase the risk of kidney failure and heart failure.[68]
Pain is typically treated with opioids, varying in potency based on the severity of symptoms. When opioids are used for prolonged periods, drug tolerance, chemical dependency, and addiction may occur. Opiate addiction is not typically a concern, since the condition is not likely to ever completely disappear. Thus, lifelong treatment with opioids is fairly common for chronic pain symptoms, accompanied by periodic titration that is typical of any long-term opioid regimen.
Intravenous Immunoglobulins (IVIGs)
Intravenous immunoglobulins may be used to control SLE with organ involvement, or vasculitis. It is believed that they reduce antibody production or promote the clearance of immune complexes from the body, even though their mechanism of action is not well-understood.[75] Unlike immunosuppressives and corticosteroids,IVIGs do not suppress the immune system, so there is less risk of serious infections with these drugs.[76]
Lifestyle changes
Avoiding sunlight is the primary change to the lifestyle of SLE sufferers, as sunlight is known to exacerbate the disease, as is the debilitating effect of intense fatigue. These two problems can lead to patients becoming housebound for long periods of time. Drugs unrelated to SLE should be prescribed only when known not to exacerbate the disease. Occupational exposure to silica, pesticides and mercury can also make the disease worsen.[35]
Renal transplantation
Renal transplants are the treatment of choice for end-stage renal disease, which is one of the complications of lupus nephritis, but the recurrence of the full disease is common in up to 30% of patients.[77]
Antiphospholipid syndrome
Antiphospholipid syndrome is also related to the onset of neural lupus symptoms in the brain. In this form of the disease the cause is very different from lupus: thromboses (blood clots or "sticky blood") form in blood vessels, which prove to be fatal if they move within the blood stream.[62] If the thromboses migrate to the brain, they can potentially cause a stroke by blocking the blood supply to the brain.
If this disorder is suspected in patients, brain scans are usually required for early detection. These scans can show localized areas of the brain where blood supply has not been adequate. The treatment plan for these patients requires anticoagulation. Often, low-dose aspirin is prescribed for this purpose, although for cases involving thrombosis anticoagulants such as warfarin are used.[78]
Management of pregnancy
Further information: Systemic lupus erythematosus and pregnancy
While most infants born to mothers who have SLE are healthy, pregnant mothers with SLE should remain under medical care until delivery. Neonatal lupus is rare, but identification of mothers at highest risk for complications allows for prompt treatment before or after birth. In addition, SLE can flare up during pregnancy, and proper treatment can maintain the health of the mother longer. Women pregnant and known to have anti-Ro (SSA) or anti-La antibodies (SSB) often have echocardiograms during the 16th and 30th weeks of pregnancy to monitor the health of the heart and surrounding vasculature.[66]
Contraception and other reliable forms of pregnancy prevention is routinely advised for women with SLE, since getting pregnant during active disease was found to be harmful. Lupus nephritis was the most common manifestation.
Prognosis
In the 1950s, most people diagnosed with SLE lived fewer than five years. Today, over 90% now survive for more than ten years, and many live relatively asymptomatically. 80-90% can expect to live a normal lifespan.[79]
Prognosis is typically worse for men and children than for women; however, if symptoms are present after age 60, the disease tends to run a more benign course. Early mortality, within 5 years, is due to organ failure or overwhelming infections, both of which can be altered by early diagnosis and treatment. The mortality risk is fivefold when compared to the normal population in the late stages, which can be attributed to cardiovascular disease from accelerated atherosclerosis, the leading cause of death for people with SLE.[68]
To reduce potential for cardiovascular issues, high blood pressure and high cholesterol should be prevented or treated aggressively. Steroids should be used at the lowest dose for the shortest possible period, and other drugs that can reduce symptoms should be used whenever possible.[68] High serum creatinine, hypertension,nephrotic syndrome, anemia and hypoalbuminemia are poor prognostic factors.[80]
The ANA is the most sensitive screening test for evaluation, whereas anti-Sm (anti-Smith) is the most specific. The dsDNA (double-stranded DNA) antibody is also fairly specific and often fluctuates with disease activity; as such, the dsDNA titre is sometimes useful to monitor disease flares or response to treatment.[81]
Epidemiology
The rate of SLE varies considerably between countries, ethnicity, gender, and changes over time.[82] In the United States, one estimate of the prevalence of SLE is 53 per 100,000;[82] other estimates range from 322,000 to over 1 million.[83] In Northern Europe the rate is about 40 per 100,000 people.[84] SLE occurs more frequently and with greater severity among those of non-European descent.[83] That rate has been found to be as high as 159 per 100,000 among those of Afro-Caribbean descent.[82]
While the onset and persistence of SLE can show disparities between genders, socioeconomic status also plays a major role. Women with SLE and of lower socioeconomic status have been shown to have higher depression scores, higher body mass index, and more restricted access to medical care than women of higher socioeconomic statuses with the illness. SLE patients had more self-reported anxiety and depression scores if they were from a lower socioeconomic status[85]
There are assertions that race and SLE are correlated. However, a 2010 review of studies which correlate race and SLE identified several sources of systematic and methodological error, indicating that the connection between race and SLE may be spurious.[86] For example, studies show that social support is a modulating factor which buffers against SLE-related damage and maintains physiological functionality.[86] Studies have not been conducted to determine whether patients of different racial backgrounds receive differing levels of social support.[86] If there is a difference, this could act as a confounding variable in studies correlating race and SLE. Another caveat to note when examining studies about SLE is that symptoms are often self-reported. This process introduces additional sources of methodological error. Studies have shown that self-reported data is affected by more than just the patients experience with the disease- social support, level of helplessness, and abnormal illness-related behaviors also factor into a self-assessment. Additionally, other factors like the degree of social support which a patient receives, socioeconomic status, health insurance, and access to care can contribute to an individual’s disease progression.[86] [87] It is important to note that racial differences in lupus progression have not been found in studies that control for the socioeconomic status [SES] of participants.[86][88] Studies that control for the SES of its participants have found that non-white patients have more abrupt disease onset compared to white patients, and that their disease progresses more quickly. Non-white patients often report more hematological, serosal, neurological, and renal symptoms. However, the severity of symptoms and mortality are both similar in white and non-white patients. Studies that report different rates of disease progression in late stage SLE are most likely reflecting differences in socioeconomic status and the corresponding access to care.[86] The patients who receive medical care often have accrued less disease-related damage, and are less likely to be below the poverty line.[88] Additional studies have found that education, marital status, occupation, and income create a social context which contributes to disease progression.[86]
SLE, like many autoimmune diseases, affects females more frequently than males, at a rate of almost 9 to 1.[82] This is due to the presence of two X chromosomes in the female as opposed to the one X chromosome present in males. The X chromosome carries immunological related genes, which can mutate and contribute to the onset of SLE. The Y chromosome has no identified mutations associated with autoimmune disease.[89]
Hormonal mechanisms could explain the increased incidence of SLE in females. The onset of SLE could be attributed to the elevated hydroxylation of estrogen and the abnormally decreased levels of androgens in female patients. In addition, differences in GnRH signalling have also shown to contribute to the onset of SLE. While females are more likely to relapse than males, the intensity of these relapses is the same for both sexes[90]
In addition to hormonal mechanisms, specific genetic influences found on the X chromosome may also contribute to the development of SLE. Studies indicate that the X chromosome can determine the levels of sex hormones. A study has shown an association between Klinefelter’s syndrome and SLE. XXY males with SLE have an abnormal X-Y translocation resulting the in the partial triplication of the PAR1 gene region.[91]
The incidence of SLE in the United States increased from 1.0 in 1955 to 7.6 in 1974. Whether the increase is due to better diagnosis or to increasing frequency of the disease is unknown.[82]
The global prevalence of SLE is approximately 20-70/100,000 people.The global age distribution of SLE ranges from ages 2–80+. In females, incidence is highest between 45-64 year of age. The lowest overall incidences exist in Iceland and Japan. The highest incidences exist in US and France. However, there is no sufficient evidence to conclude that SLE is less common in some countries compared to others, since there is significant environmental variability in these countries. For example, different countries receive different levels of sunlight, and exposure to UV rays affects dermatological symptoms of SLE. Certain studies hypothesize that a genetic connection exists between race and lupus which affects disease prevalence. If this is true, the racial composition of countries affects disease, and will cause the incidence in a country to change as the racial makeup changes. In order to understand if this is true, countries with largely homogenous and racially stable populations should be studied to better understand incidence.[92]
History
The history of SLE can be divided into three periods: classical, neoclassical, and modern. In each period, research and documentation advanced the understanding and diagnosis of SLE, leading to its classification as an autoimmune disease in 1851, and to the various diagnostic options and treatments now available to SLE patients. The advances made by medical science in the diagnosis and treatment of SLE has dramatically improved the life expectancy of a person diagnosed with SLE.[93]
Etymology
There are several explanations ventured for the term lupus erythematosus. Lupus is Latin for wolf,[94] and "erythro" is derived from ερυθρός, Greek for "red." All explanations originate with the reddish, butterfly-shaped malar rash that the disease classically exhibits across the nose and cheeks.
- In various accounts, some doctors thought the rash resembled the pattern of fur on a wolf's face. More likely is that it is derived from the similarity in distribution to lupus vulgaris or chronic facial tuberculosis where the lesions are ragged and punched out and are said to resemble the bite of a wolf.
- Another account claims that the term "lupus" did not come from Latin directly, but from the term for a French style of mask that women reportedly wore to conceal the rash on their faces. The mask is called a "loup," French for "wolf."
Classical period
The classical period began when the disease was first recognized in the Middle Ages. The term lupus is attributed to 12th-century Italian physician Rogerius Frugard, who used it describe ulcerating sores on the legs of patients.[95] No formal treatment for the disease existed and the resources available to physicians to relieve the suffering of their patients was limited.[96]
Neoclassical period
The neoclassical period began in 1851 when the skin disease now known as discoid lupus was documented by French physician, Pierre Cazenave. Cazenave termed the illness lupus and added the word erythematosus to distinguish this disease from other illnesses that affected the skin but were also infectious.[97]Cazenave observed the disease in several patients and made very detailed notes to assist others in itsdiagnosis. He was one of the first to document that lupus affected adults from adolescence into the early thirties and that the facial rash is its most distinguishing feature.[98]
Research and documentation of the disease continued in the neoclassical period with the work of Ferdinand von Hebra and his son-in-law, Moritz Kaposi. They documented the physical effects of lupus as well as some insights into the possibility that the disease caused internal trauma. von Hebra observed that lupus symptoms could last many years and that the disease could go "dormant" after years of aggressive activity and then re-appear with symptoms following the same general pattern. These observations led Hebra to term lupus a chronic disease in 1872.[99]
Kaposi observed that lupus assumed two forms: the skin lesions (now known as discoid lupus) and a more aggravated form that affected not only the skin but also caused fever, arthritis, and other systemic disorders in patients.[100] The latter also presented a rash confined to the face, appearing on the cheeks and across the bridge of the nose; he called this the "butterfly rash". Kaposi also observed those patients who developed the "butterfly rash" (or malar rash) often were afflicted with another disease such as tuberculosis, anemia, or chlorisis which often caused death.[98] Kaposi was one of the first persons to recognize what is now termed systemic lupus erythematosus in his documentation of the remitting and relapsing nature of the disease and the relationship of skin and systemic manifestations during disease activity.[101]
The 19th century's research into lupus continued with the work of Sir William Osler who, in 1895, published the first of his three papers about the internal complications of erythema exudativum multiforme. Not all the patient cases in his paper suffered from SLE but Osler's work expanded the knowledge of systemic diseases and documented extensive and critical visceral complications for several diseases including lupus.[98] Noting that many patients with lupus had a disease that not only affected the skin but many other organs in the body as well, Osler added the word "systemic" to the term lupus erythematosus to distinguish this type of disease from discoid lupus erythematosus.[102] Osler's second paper noted that reoccurrence is a special feature of the disease and that attacks can be sustained for months or even years. Further study of the disease led to a third paper, published in 1903, documenting afflictions such as arthritis, pneumonia, the inability to form coherent ideas, delirium, and central nervous system damage as all affecting patients diagnosed with SLE.[98]
Modern period
The modern period, beginning in 1920, saw major developments in research into the cause and treatment of discoid and systemic lupus. Research conducted in the 1920s and 1930s led to the first detailed pathologic descriptions of lupus and demonstrated how the disease affected the kidney, heart, and lung tissue.[103] A major breakthrough was made in 1948 with the discovery of the LE cell (the lupus erythematosus cell—a misnomer, as it occurs with other diseases as well). Discovered by a team of researchers at the Mayo Clinic, they discovered that the white blood cells contained the nucleus of another cell that was pushing against the white's cell proper nucleus.[104] Noting that the invading nucleus was coated with anti-body that allowed it to be ingested by a phagocytic or scavenger cell, they named the antibody that causes one cell to ingest another the LE factor and the two-nuclei cell result the LE cell.[105] The LE cell, it was determined, was a part of an anti-nuclear antibody (ANA) reaction; the body produces antibodies against its own tissue. This discovery led to one of the first definitive tests for lupus for LE cells are found in approximately 60% of all people diagnosed with lupus.[106] (Note: The LE cell test is rarely performed as a definitive lupus test today as LE cells do not always occur in lupus patients and can occur in individuals with other autoimmune diseases. Their presence, however, can be helpful in establishing a diagnosis but is no longer indicates a definitive SLE diagnosis)
The discovery of the LE cell led to further research and this resulted in more definitive tests for lupus. Building on the knowledge that those with SLE had auto-antibodies that would attach themselves to the nuclei of normal cells, causing the immune system to send white blood cells to fight off these "invaders", a test was developed to look for the anti-nuclear antibody (ANA) rather than the LE cell specifically. This ANA test was easier to perform and led not only to a definitive diagnosis for lupus but also many other related diseases. This discovery led to the development of what are now known as autoimmune diseases.[107]
To ensure that the patient has lupus and not another autoimmune disease, the American College of Rheumatology (ACR) established a list of clinical and immunologic criteria that, in any combination, point to SLE. The criteria include symptoms that the patient can identify (e.g. pain) and things that a physician can detect in a physical examination and through laboratory test results. The list was originally compiled in 1971, initially revised in 1982, and further revised and improved in 2009.[108]
Medical historians have theorized that people with porphyria (a disease that shares many symptoms with SLE) generated folklore stories of vampires and werewolves, due to the photosensitivity, scarring, hair growth, and porphyrin brownish-red stained teeth in severe recessive forms of porphyria (or combinations of the disorder, known as dual, homozygous, or compound heterozygous porphyrias).[109]
Useful medication for the disease was first found in 1894, when quinine was first reported as an effective therapy. Four years later, the use of salicylates in conjunction with quinine was noted to be of still greater benefit. This was the best available treatment until the middle of the twentieth century, when Hench discovered the efficacy of corticosteroids in the treatment of SLE.[109]
Notable cases
- Charles Kuralt, former anchor of CBS Sunday Morning, died of SLE complications in 1997.[110]
- Donald Byrne, American chess player who died from SLE complications in 1976.[111]
- Ferdinand Marcos, former Philippine president, died of SLE complications in 1989.[112]
- Flannery O'Connor, American fiction writer who died of SLE complications in 1964.[113]
- Hugh Gaitskell, British politician who died of SLE complications in 1963 aged 56.[114]
- Inday Ba (also known as N'Deaye Ba), a Swedish-born actress who died from SLE complications at age 32.[115]
- J Dilla (also known as Jay Dee), a hip-hop producer and beat maker who died of SLE complications in 2006.[116]
- Caroline Dorough-Cochran, sister of Howie D. of the Backstreet Boys, died of SLE complications. He founded the Dorough Lupus Foundation in her memory.
- Lady Gaga has been tested borderline positive for SLE, however she claims not to be affected by the symptoms yet. The revelations caused considerable dismay amongst her fans, leading to Gaga herself addressing the matter in an interview with Larry King,[117] saying she hopes to avoid symptoms by maintaining a healthy lifestyle.[118] However, she has recently been experiencing complications from synovitis that are possibly related to her lupus.[119]
- Lauren Shuler Donner, American movie producer.[120]
- Lucy Vodden, inspiration for the Beatles song Lucy In The Sky With Diamonds. Meeting Vodden later in life and realizing her illness, Julian Lennon campaigned to raise awareness of lupus and following Vodden's death in 2009 has been an active member of the Lupus Foundation of America.[121][122]
- Louisa May Alcott, American author best known for her novel Little Women, has been suggested to have had SLE.[123]
- Mary Elizabeth McDonough, American actress; blames her SLE on leaky silicone breast implants.[124]
- Mercedes Scelba-Shorte, America's Next Top Model Season Two runner-up and model.[125]
- Michael Jackson suffered from both SLE and vitiligo.[126] Diagnosed in 1986, and confirmed by his dermatologist, Arnold Klein, who presented legal documents during court depositions.
- Michael Wayne, Hollywood director, and producer, part owner of Batjac Productions, son of legendary actor John Wayne, died of heart failure resulting from SLE complications in 2003.[127]
- Pumpuang Duangjan, "Queen of Thai Country Music"
- Ray Walston, character actor who died of SLE complications in 2001 after a six-year battle with the disease.[128]
- Sophie Howard, British glamour model[129]
- Teddi King, American singer, died of SLE complications in 1977.[130]
- Tim Raines, former major league baseball player[131]
- Toni Braxton was hospitalized in Los Angeles in December 2012 because of "minor health issues" related to Lupus.[132]
Research
Since lupus is considered to be currently incurable, current research is being geared towards finding a possible cause, a cure, and more effective treatment plans to extend and increase the quality of life for lupus patients.
A study called BLISS-76 tested the drug, Belimumab (HGS1006, LymphoStat-B ), a fully human monoclonal anti-BAFF (or anti-BLyS) antibody.[70] stimulates and extends the life of B lymphocytes, which produce antibodies against foreign and self cells.[133] The drug, branded Benlysta, was approved by the FDA in March 2011.[69]




No comments:
Post a Comment